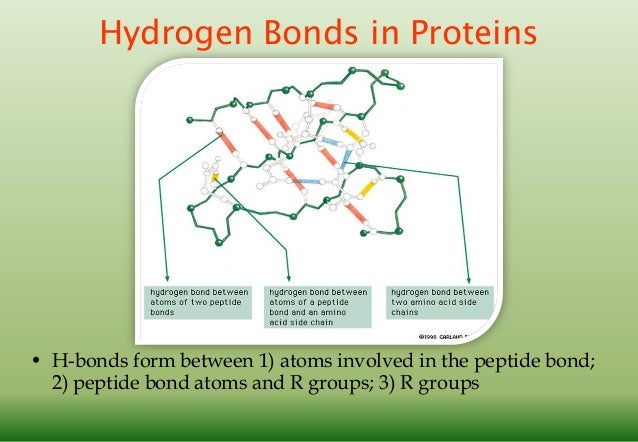
Click on the Jmol frank, in the main menu which opens click on Console, in the bottom box enter the commands: select protein calculate hbonds structure and then click Run. One of the new features of Jmol 12.0 is a command, calculate hbonds structure, which determines and displays the hbonds in helices, sheets and turns. PyMOL likewise displays "polar contacts" using dashed bonds between the involved atoms, leaving further assessment of hydrogen bonding to the user. Protein Explorer and FirstGlance in Jmol (see applet below) have Contacts dialogs that show putatively hbonded donors and acceptors based simply on the chemical elements and interatomic distances. 12, free molecular visualization programs displayed potential donor-acceptor pairs, deeming them "putatively" hbonded because determining the positions of hbonds with high confidence required expert and detailed examination of the donor-acceptor chemistry and geometry. Jmol can now display hydrogen bonds, as can several other software packages.īefore the availability of Jmol ver. 14).Ī common cutoff donor-to-acceptor distance for energetically significant hydrogen bonds in proteins is 3.5 Å (see legend to Table 6 in Kajander et al. The hydrogen atoms in moderate hbonds often do not lie on the straight line connecting the donor to acceptor, so donor-acceptor distance slightly underestimates the length of the hbond (Jeffrey, p. Strong hbonds require moieties or conditions that are rare within proteins. Most hbonds in proteins are in the moderate category. Energies are given as 40-14, 15-4, and <4 kcal/mol respectively. Jeffrey (page 12) categorizes hbonds with donor-acceptor distances of 2.2-2.5 Å as "strong, mostly covalent", 2.5-3.2 Å as "moderate, mostly electrostatic", and 3.2-4.0 Å as "weak, electrostatic". The mean donor-acceptor distances in protein secondary structure elements are close to 3.0 Å, as are those between bases in Watson-Crick pairing (Jeffrey, pp. The C-H bond has insufficient charge separation, and C in amino acids has no lone electron pair. Lacking hydrogens, these latter cannot serve as donors.Ĭarbon atoms lack sufficient electronegativity to serve as energetically significant hydrogen bond donors or acceptors. So can those on carbonyl oxygens =O (as in the protein main chain) or nitrogens with three covalent bonds =N- (as in the sidechains of His, Trp, or in nucleotide bases). The lone electron pairs on these same donors can serve as hbond acceptor sites. the sidechains of Ser, Thr, Tyr), HOH, and the nitrogen in -NH3+ (as in the sidechains of Lys, Arg) or -NH- (as in the main chain peptide bond, and the sidechains of Trp, His, Arg, and nucleotide bases). Typical donor atoms are the oxygens in -OH (e.g. Hydrogen bonds ("hbonds") are non-covalent bonds that occur when a donor atom donates its covalently bonded hydrogen atom to an electronegative acceptor atom. The dotted hydrogen bond is non-covalent and variable in length, ~1.5-2.5 Å. In this example, the N-H bond is covalent and fixed in length. Distances shown in Å are typical for those found in proteins. These results suggest that backbone hydrogen bonding is critical for the assembly of amyloid fibrils.Elements: C, H, N, O.A hydrogen bond (dotted white line) between a nitrogen donor and an oxygen acceptor. Abeta16-20e also inhibits the aggregation of the Abeta1-40 peptide and disassembles preformed Abeta1-40 fibrils. Analytical ultracentrifugation experiments demonstrate that this ester peptide, Abeta16-20e, is predominantly monomeric under solution conditions, unlike the fibril-forming Abeta16-20 peptide. The ester bonds were incorporated in an alternating fashion so that the peptide presents two unique hydrogen bonding faces when arrayed in an extended, beta-strand conformation one face of the peptide has normal hydrogen bonding capabilities, but the other face is missing amide protons and its ability to hydrogen bond is severely limited. In this paper, we report that interfering with the backbone hydrogen bonding of an amyloidgenic peptide (Abeta16-20) by replacing amide bonds with ester bonds prevents the aggregation of the peptide. Evidence also suggests that the macromolecular assemblies of peptides and proteins in amyloid fibrils are stabilized by intermolecular beta-sheets. A number of cytokines and the HIV Protease, for example, dimerize through beta-sheet motifs. Protein-protein interactions are frequently mediated by stable, intermolecular beta-sheets.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
